 
In this arrangement, Cl- ions are present at the corners and at the centre of each face of the cube. (i) Chloride ions (In a typical unit cell) are arranged in cubic close packing (ccp). Thus the salient features of this structure are as follows: 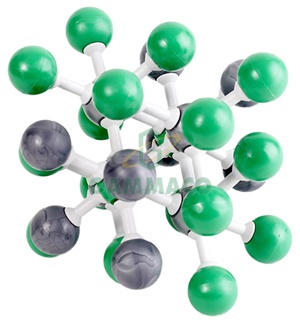
The radius ratio of 0.524 for NaCl suggest an octahedral void. The radii of Na+ and Cl- ions 95 pm and 181 pm giving the radius ratio of 0.524 The number of sodium ions is equal to that of Cl- ions. The sodium chloride structure is composed of Na+ and Cl- ions. Sodium chloride (Rock salt) type structure (A) ZnS type (B) NaCl types (C) CsCl typesġ. Ionic compounds of the type AB have three types of crystalline structures. (c) When the anions (B -2) are constituting space lattice and all the tetrahedral voids are occupied by the cations (A+), then the formula of the solid crystal will be A2B. (b) Similarly, if half of the tetrahedral voids are occupied by cations, then the formula of the solid crystal becomes A+B. (a) If the anions (B-) constitute the crystal lattice and all octahedral voids are occupied by cations (A+), then the formula of the ionic solid is AB. From the knowledge of close packed structures and the voids developed there in, we can have an idea about the structures of simple ionic compounds.Īmong the two ions, constituting the binary compounds, the anions usually constitute the space lattice with hcp or ccp type of arrangements whereas the cations, occupy the interstitial voids Simple ionic compounds are of two types i.e. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
